Everything in the universe which we see as matter lies in the atom since its structure defines the physical and chemical properties of elements
What is the smallest particle of matter. What is a substance made from one kind of atom. What do you call one or two letters used to represent an. The smallest particle of matter is called. Tags: Question 3. The length of an atom's nucleus. The center of a compound. Another name for mixtures that go together.
- An atom can be defined as the smallest particles of an element which can take part in a chemical reaction. This note provides information about compound, atom, molecule, molecular formula and change in matter.
- Quarks represent the smallest known subatomic particles. These building blocks of matter are considered the new elementary particles, replacing protons, neutrons and electrons as the fundamental particles of the universe. There are six types, called flavors of quarks: up, down, charm, strange, top and bottom.
- An atom is the smallest unit of matter that retains all of the chemical properties of an element. Learn about the structure of the atom, and how atoms make up matter. If you're seeing this message, it means we're having trouble loading external resources on our website.
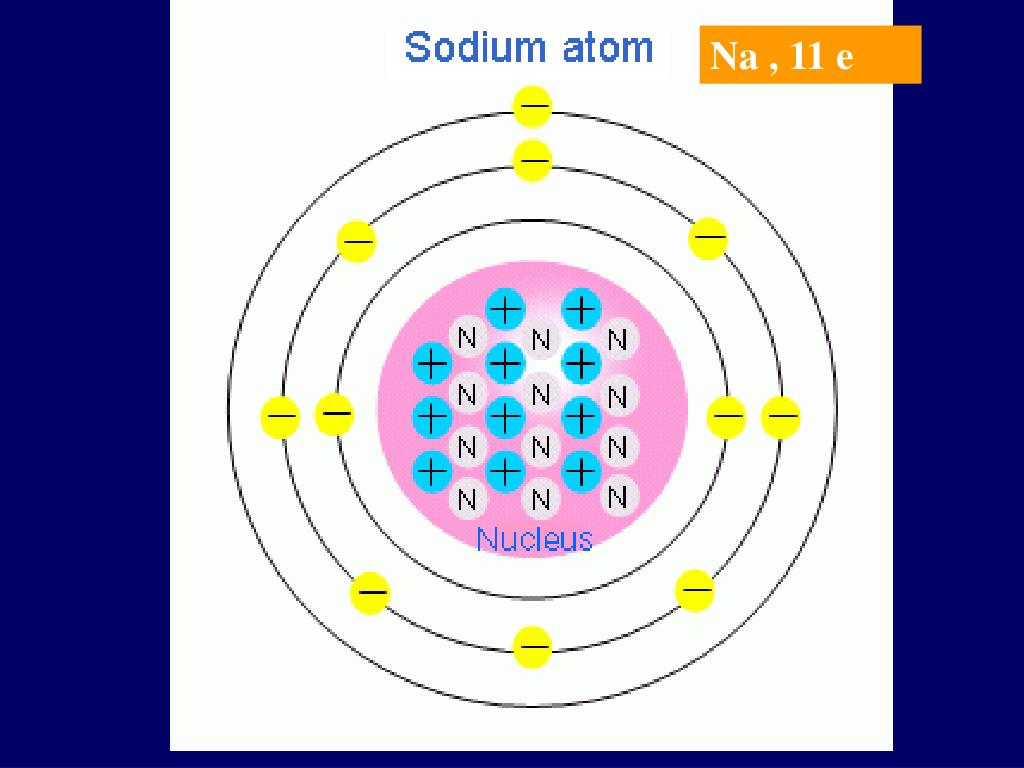
An atom is the smallest part of matter that has the properties of a chemical element. The atom is composed of a nucleus and electrons linked to the nucleus. The nucleus is made of one or more protons and, typically, a similar number of neutrons; protons and neutrons are called nucleons.
The protons of the atomic nucleus are united by very strong bonds of strong nuclear force in which has a large amount of energy at a very small distance.
Origin of word Atom
The origin of the word atom comes from Greek and means indivisible. The conception of indivisible comes from antiquity when it was believed that the atom was the smallest element that could exist. However, we now know that the atom is made up of still smaller particles: subatomic particles.
The Smallest Particle Of Matter Still Retaining The Characteristics
What does an atom made of?
The atom contains protons, neutrons, and electrons. electron is the lightest particle of how many make up the atom. It has a negative electric charge, whose magnitude is defined as the elementary electric charge, and it is ignored if it has a substructure, so it is considered an elementary particle. The mass of a proton is 1836 times greater than the mass of the electron. The charge of the proton is positive. The neutron has a mass 1839 times the mass of the electron.
The proton and the neutron are not elementary particles but are a composite of other particles called quarks. Subatomic particles constitute a bound state of quarks u and d. A proton contains two quarks u and a quark d, while the neutron contains two d and u, consistent with the charge of both. The quarks are held together by the strong nuclear force, mediated by gluons.
Smallest Piece Of Matter
if you are interested in the science go through the bellow links
